With a complete set of equipments, a flexible production process, multiple GMP compliant workshops available, it can satisfy with the requirements of virus production for pre-clinical and clinical trial studies.
Viral
vector
With more than 10 years of devotion to the field of oncolytic viruses, Binhui Bio has accumulated a wealth of experience in viral vector construction, viral and cell bank establishment, process development, lab and pilot scale production, quality control, and IND application in China and the United States, etc., and has developed a mature service platform for virus vector technology that can provide R&D and production for herpes simplex virus, adenovirus, poxvirus, lentivirus, retrovirus, and other virus vectors.
Service
contentBank Establishment for Cells/Viral Strains
Internal Checking of Cell/Viral Strain Bank
Analysis Method Development
Analysis Method Verification
Culture Process Development
Bioreactor Process Development
Virus Purification Process Development
Formulation Process Development
Clinical Batch of Production
Sample Stability Study
IND Document Writing
Production
Process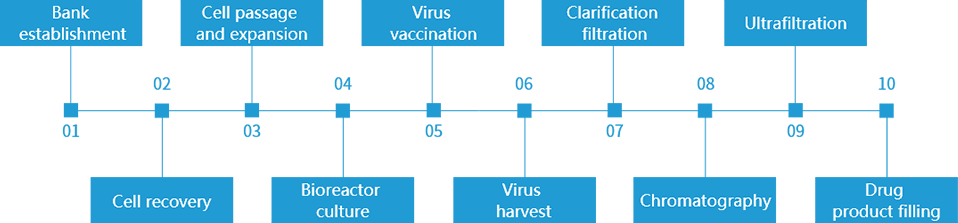
Service
featuresWith a complete set of equipments, a flexible production process, multiple GMP compliant workshops available, it can satisfy with the requirements of virus production for pre-clinical and clinical trial studies.
Three-level bank establishment for virus seed bank and cell bank in GMP compliant workshop, suspended or adherent cell culture, 3L-50L-100L scale-up production.
Disposable and aseptic production process, A-level aseptic isolator filling system, personnel and materials are separated to minimize contamination risk.
Integrated QC system, strict monitoring of the production process, product release, and complete and traceable CMC original records.
High yield, less impurity residue, and low production and purification cost.
Rich experience in different scale of production and IND application. Several viral vector drugs were successfully approved for Sino-US clinical trial application.
Viral Vector Service Manual
Download